Get involved in research
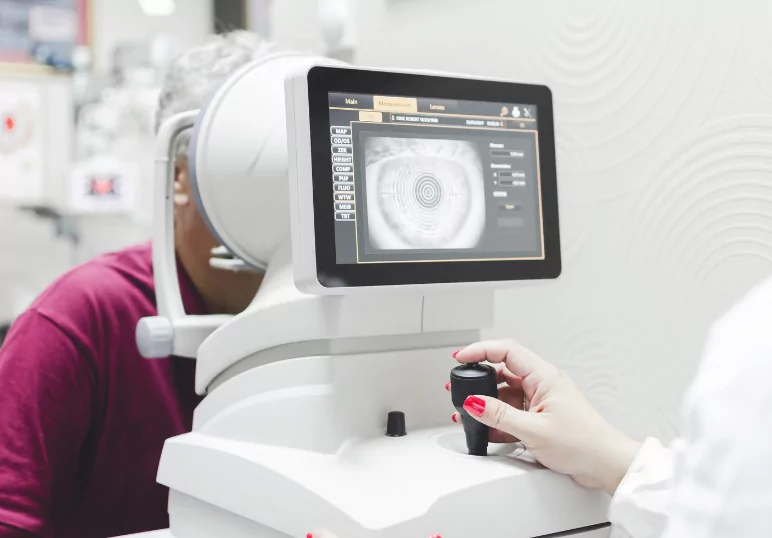
Auckland Eye conduct clinical trials to evaluate new treatments and interventions across a wide range of eye conditions. By participating in a clinical trial you are helping to advance medical knowledge and contributing to the development of new medicines and devices which may benefit others in the future.
Current Clinical Trials

We are currently recruiting participants for the CoMpass Study, which aims to evaluate the safety and effectiveness of an investigational treatment called bel-sar when it is given to people with Small Choroidal Melanoma or Indeterminate Lesions (types of eye cancer), compared to a sham treatment.
How Does Bel-sar Work?
Bel-sar is a light-activated liquid which aims to target and destroy tumor cells in the eye without affecting the healthy cells. It is injected into the eye using an investigational microinjector. A laser is then shone onto the tumor in your eye to activate bel-sar.
Who Can Participate?
Adults aged 18 and older with suspected or confirmed eye cancer (Small Choroidal Melanoma or Indeterminate Lesions) who have not received prior treatment for it.
What does the trial involve?
If eligible, participants will be randomly assigned to one of three groups: a higher dose of the investigational treatment bel-sar, a lower dose of bel-sar, or a sham treatment. Overall participants have a 60% chance of receiving the active drug. The study will take place at Auckland Eye over approximately 104 weeks (2 years), involving around 29 visits for assessments such as blood tests, vision exams, and comprehensive eye examinations.
All study treatment and study-related care will be provided at no cost, and participants will be reimbursed for reasonable transportation expenses.
Contact Us
If you are interested in learning more about this study, please email research@aucklandeye.co.nz or phone (09) 529 2480.

The i-SIGHT2 AMD Study is actively recruiting participants to evaluate the i-Lumen AMD microcurrent electrical stimulation device. This research aims to determine the safety and effectiveness of the device, which delivers low-level electrical stimulation to the eyelid, potentially improving vision and slowing the progression of dry age-related macular degeneration (AMD).
Who Can Participate?
Adults aged 60 and older with vision loss caused by (dry) age-related macular degeneration (AMD) in at least one of your eyes.
What does the trial involve?
If eligible, participants will be randomly assigned to receive either active microcurrent study treatment or sham (inactive) study treatment. Participants will have a 66.6% chance of receiving the active device.
The study will take place at Auckland Eye over approximately 15 months involving 22 visits for various assessments, including vision and comprehensive eye exams.
All treatment and study-related care will be provided at no cost, and participants will be reimbursed for reasonable transportation expenses.
Contact Us
If you are interested in learning more about this study, please email research@aucklandeye.co.nz or phone (09) 922 6734.
We’re recruiting for a clinical trial investigating a potential new eyedrop for the treatment of pterygium (this is a wedge-shaped growth that extends onto the cornea of the eye).
Who can take part?
- Adults aged 18 years and over
- Have a diagnosis of pterygium
- Are in good general health
What does the trial involve?
After an initial screening visit, eligible participants will be randomised to receive either one of two different dose strengths of the study eyedrop or a placebo. The study is being conducted at Auckland Eye, based near Newmarket in Remuera, and will require approximately 12 visits over 24-months. Participants may have the opportunity to switch to the higher dose strength at the 18-month visit. The treatment and study-related care will be provided at no cost. Participants will be reimbursed for reasonable transport costs to attend study visits.
ClinicalTrials.gov ID: NCT05456425
Click here to read the Participant Information Sheet.
Contact Us
If you are interested in learning more about this study, please contact Jas Walia via email research@aucklandeye.co.nz or phone (09) 922 6734 / 021 192 1622.
Approved by the NZ Health & Disability Ethics on 21Nov2023 [Ref: 2023 FULL 18325]

We are recruiting for a clinical trial for people who have glaucoma or ocular hypertension and need cataract surgery.
Contact Us
If you are interested in learning more about this study, please email research@aucklandeye.co.nz or phone (09) 922 6734.

· Are you currently using eyedrops for glaucoma?
· Are you between 35-85 years of age?
· Are you interested in safely stopping your eyedrops?
If you answered yes to all the above, you may be eligible to take part in a clinical research study. We are comparing two different medical devices implanted in the eye to reduce eye pressure in people with glaucoma. The study is being conducted at Auckland Eye, based near Newmarket in Remuera, and will require 13-16 visits over 24-months.
The device and study-related care will be provided at no cost. You will be reimbursed for reasonable transport costs to attend study visits.
Contact Us
If you are interested in learning more about this study, please email research@aucklandeye.co.nz or phone (09) 529 2480.
Future Clinical Trials
Exciting new studies on dry AMD are coming soon, and you may be eligible to participate.
Register here to stay informed about upcoming opportunities.
Are you interested in learning more about clinical trials for glaucoma treatment and cataract / lens replacement surgery?
Register here to receive information about upcoming studies and how you can participate.
FUTURE CLINICAL TRIALS
Join our clinical trials database
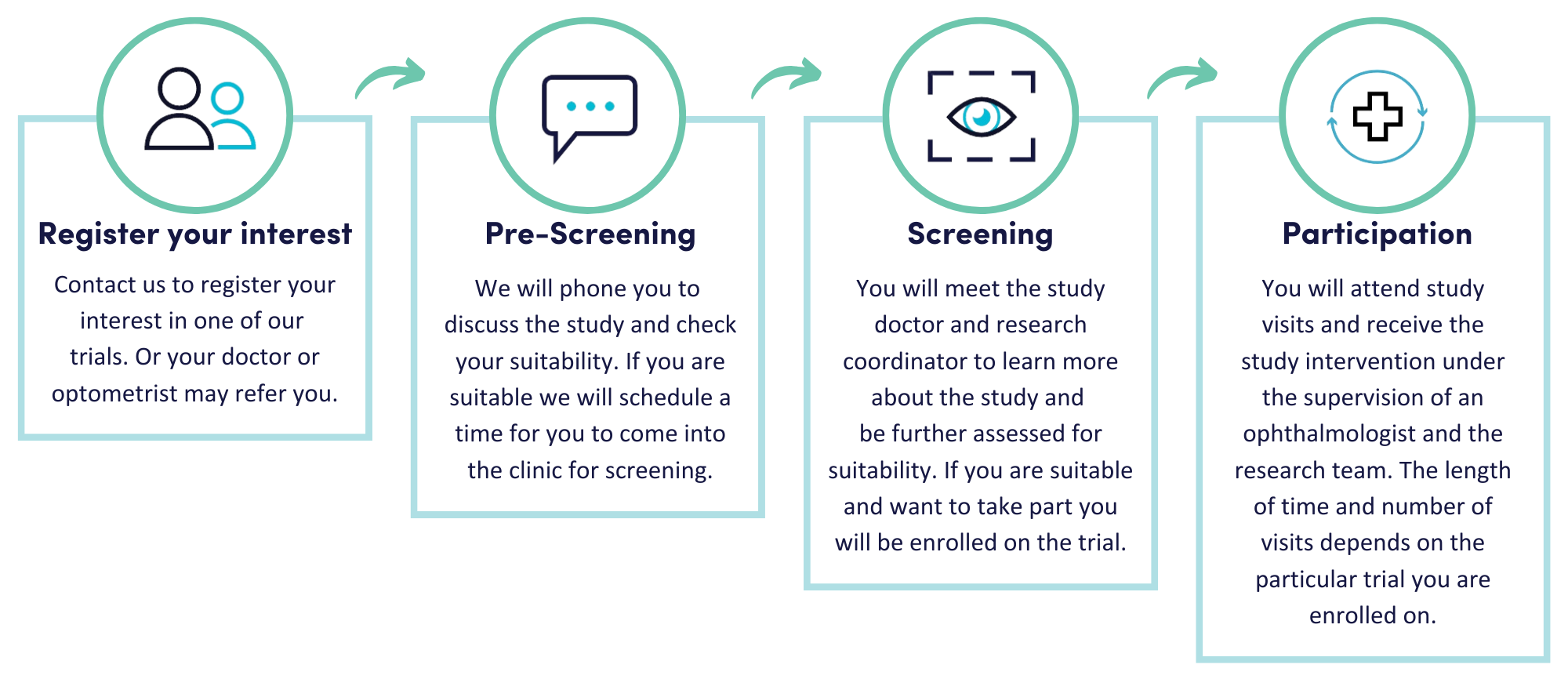
How can I participate in a clinical trial?
FAQ’s
By participating in clinical research, you may:
• Learn more about your condition/disease
• Get free access to new medicines or devices that may not otherwise be available
• Receive free regular testing and monitoring by the study team including our specialist ophthalmologists
• Contribute to the development of new medicines and devices which may benefit others in the future
Clinical trials have special eligibility criteria that determine who can take part. These criteria are used to assess your suitability for a study and make sure it is safe for you to take part, and include factors such as your age, medical conditions and treatment history.
Initially, your eye doctor or one of the research team will talk to you about the study and check whether you may be suitable. If you are interested, we will send you an information sheet which explains what the trial involves. You will then be invited to come in for a clinic visit to meet the study doctor to discuss the trial in more detail and answer any questions you may have. You will be asked to sign an Informed Consent Form and complete some screening tests to make sure you are suitable for the study and that it is safe for you to take part. We will let you know whether you are eligible and if so, you can get started on the study. You will come in for scheduled visits over the course of the study and be closely monitored by the research team.
The length of time depends on the study you are enrolled in. Some of our studies are short, lasting just a few weeks or months. Other studies can last for a year or more. You will be informed about the number of visits and how long the study lasts before you decide whether to take part.
Participation in clinical trials is voluntary. You don’t have to take part. If you change your mind about being in a study you can withdraw at any time without affecting your ongoing care.
Click here to learn about clinical trials.